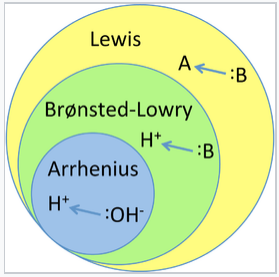
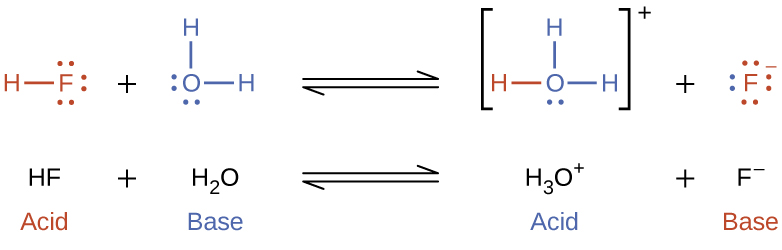
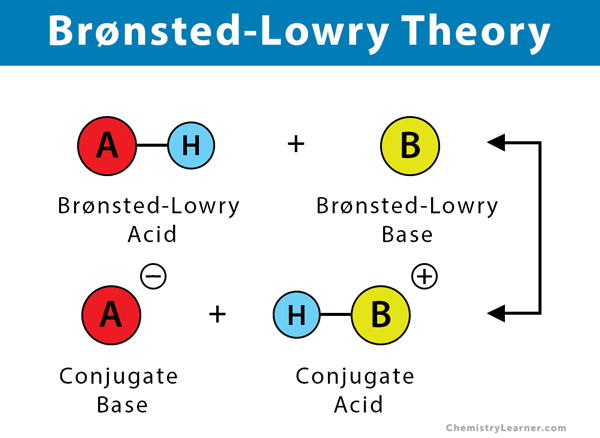
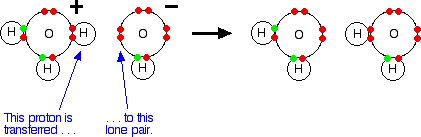
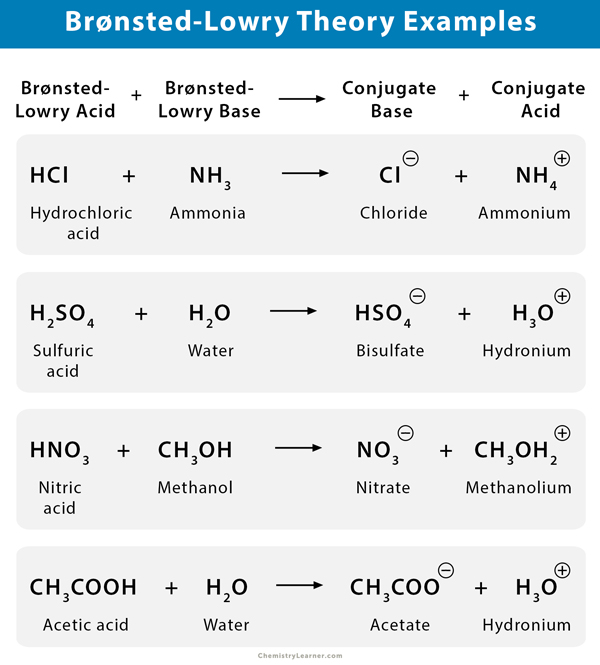
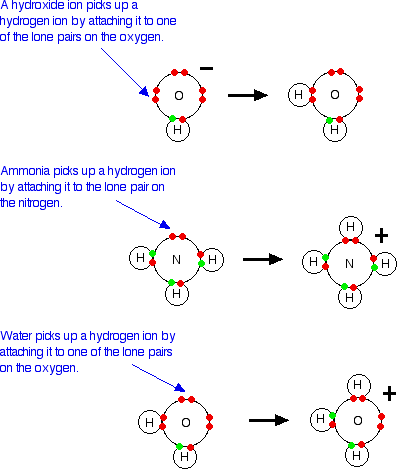
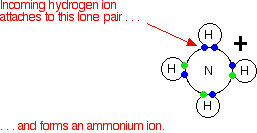
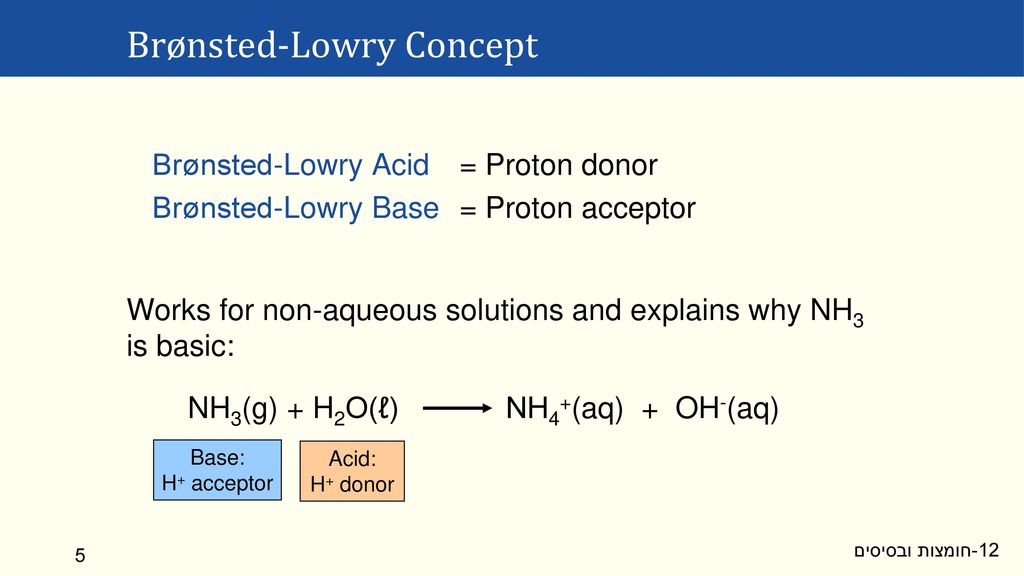
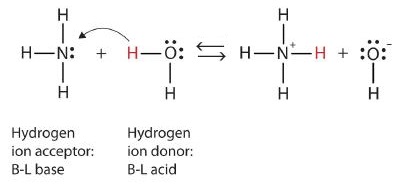
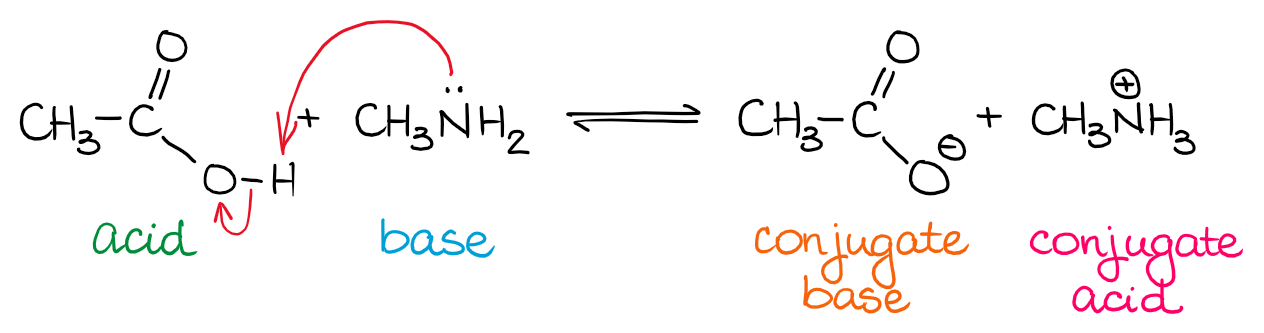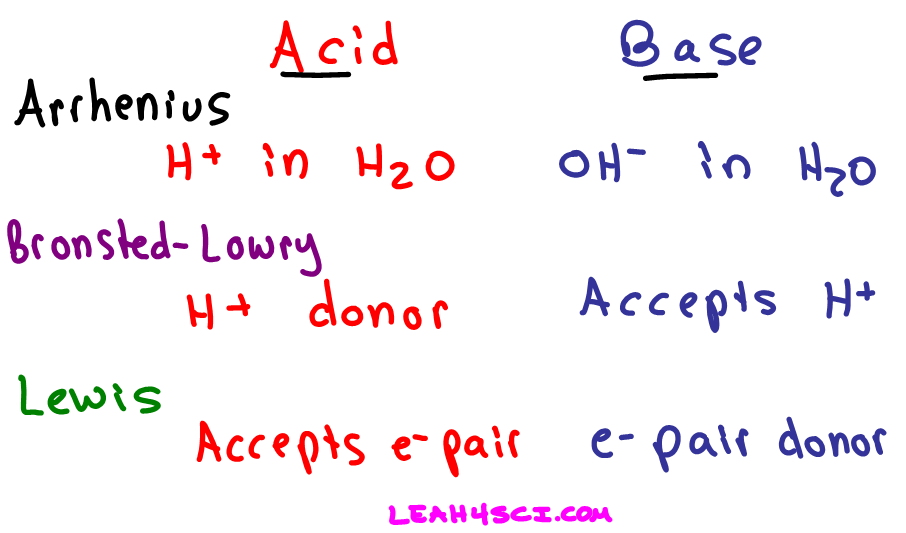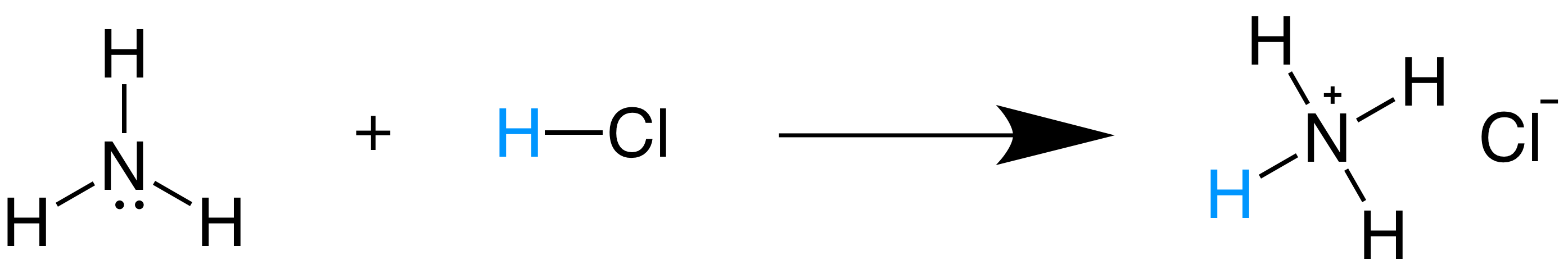What is an acid according to the Bronsted and Lowrey definitions with the help of an example? - Quora
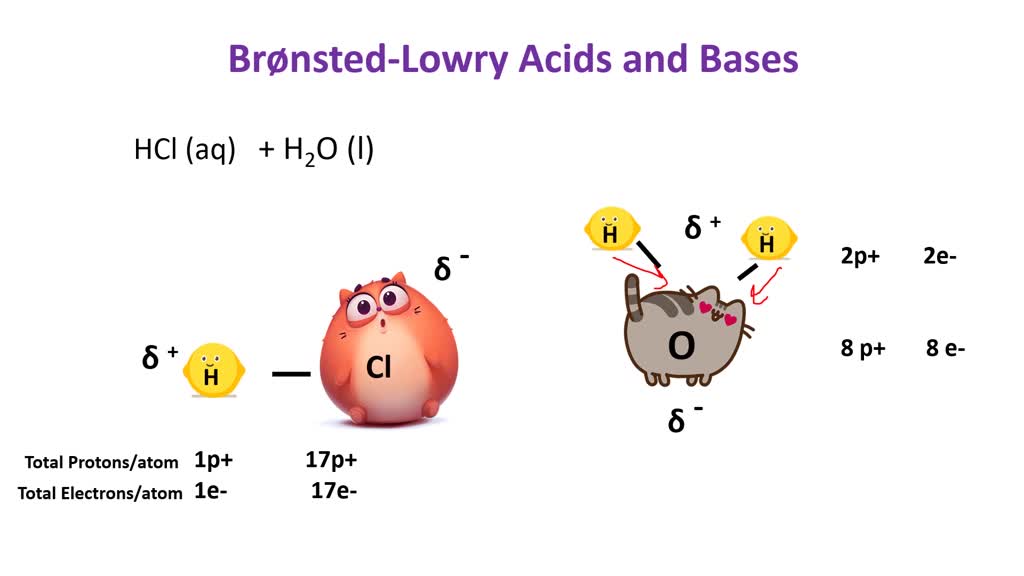
SOLVED:According to the Bronsted-Lowry model, an acid is a “proton donor” and a base is a “proton acceptor.” Explain.
Does the pH scale measure Arrhenius, Bronsted-Lowry, or neither? If it is Bronsted-Lowry, why does a lower pH mean more acidity, since an acid donates protons? - Quora